Na element charge11/6/2023 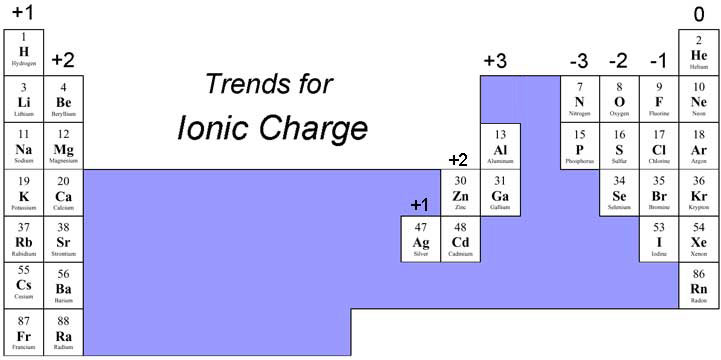
That is what makes Aluminium compounds that are ionic behave in a slightly covalent manner. Therefore, the charge will be balanced when the positive charges sum to 6 and the negative charges sum to 6. For example, if the charges of the ions are 2+ and 3, the least common multiple between 2 and 3 is 6. It might be worth knowing that very high charge density ions, particularly Al 3+ are so attractive to negative ions ( polarising) that they actually draw some of the electron densityback towards them. When balancing charges, many find it easiest to think of the least common multiple between the magnitude of charge. The same is true when comparing melting points of metals Sodium has a lower melting point than Aluminium because Na + has a lower charge density than Al 3+ - it's less charged and larger. That charge is shown as a superscript at the top, right of the symbol. Because the ion has 11 positive protons and 10 negative electrons, there is a net positive charge. The cation produced in this way, Na +, is called the sodium ion to distinguish it from the neutral element. It may be obvious that a Mg 2+ ion attracts Cl - ions more strongly than than an Na + ion would simply because of the greater charge but since it is also smaller if you do not mention charge density in an exam you will be penalised. Now it has the same number of electrons as the nearest noble gas, neon. Exam Hint Įven if you are asked to compare the strength of attraction of a 2+ and a 1+ ion you must still take the size of the ion (and hence the charge density) into account. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. This makes Na + more attractive for a Cl - ion.īut rather than write an essay explaining all this it easier to say that Na +'s small size gives it a higher charge density. Sodium is a chemical element with atomic number 11 which means there are 11 protons in its nucleus. Since Rb + is a much larger ion the 1+ charge is "spread over" a far bigger surface than it would be in Na +. (Although it really exists as sodium ions surrounded by a sea of electrons), consider it for our purposes to exist as elemental Na, which. The oxidation number of a Group 2 element in a compound is +2. The oxidation number of a Group 1 element in a compound is +1. 
The oxidation number of O in its compounds is usually -2, but it is -1 in peroxides. 
In any period, the more positive the cation the smaller its radius - giving Al 3+ a particularly high charge density responsible for many of its odd properties. The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
